Copper is a living element that changes over time, evolving alongside you. That is why you will see copper develop what is known as a patina. Copper and patina work together to create a unique finish, and the patina is a protective layer for the copper itself. While it develops as a protective featur, the greeen patina on copper, blue copper patina or brown copper patina provide a striking textural element to spaces.
If you are looking for a deep dive into how copper patina is formed and how it can elevate your home, check out this copper patina guide. Here, you can learn how copper forms patina as well as the colours it can develop, and how you can incorporate it into your home to elevate spaces.
What Is Copper Patina?
In the most simple terms, copper patina is a layer that forms over copper to protect it from exposure to the elements. This protective layer can have a blue or green hue that brings character to the copper while at the same time helping it last for decades.
How Copper Forms Patina
Copper home furnishings provide an elegant beauty because copper is a “living” metal, meaning it changes color over time. The beauty of patina copper sheets is unique among decorative materials because the changes it undergoes are unique to each home and location.
Copper Is a Part of History
The natural changes that happen to aged copper sheets and objects make them a one-of-a-kind symbol of heritage that's hard to replicate. The shades of patina that form on the surface of a copper furnishing can store the memories of meals, spills, new homes and life changes over the course of centuries.

Copper is more than a shiny metal, it is one that can be adorned with beautiful carvings that tell a story. Knowing how copper patina forms will give you a deeper appreciation for the unique patina that appears in your family's history. This knowledge will also allow you to influence what color of patina forms, and how fast it will do so.
What Is the Patina Chemical Reaction
Ever ask yourself, “Why does copper turn green?” The formation of patina on copper is similar to the formation of rust on iron. Both happen when oxygen in the air interacts with the metal atoms in the presence of water.
Copper reacts with oxygen that is in the air, resulting in copper dioxide (Equation 1). The copper dioxide then reacts with more oxygen to form copper oxide (Equation 2). This copper oxide from reaction 2 is the main culprit that will later form the colors of the patina. If sulfur is present on the surface of the copper, then the two can react to form copper sulfide, which is black (Equation 3). The greenish and blueish colors of copper’s patina come from the next set of reactions.

Different Reactions Patina Can Have
Air contains more than just the oxygen that we breathe. It contains carbon dioxide, which we breathe out, along with water vapor. The first reaction that gives patina its color is when one molecule of carbon dioxide and one molecule of water, both in the air, react with two molecules of copper oxide on the surface of the copper. The result is a compound called malachite (Equation 4), which varies in color from shades of dark green to shades of blue.
The second reaction that adds to patina’s color like blue patina is when three molecules of copper oxide from Equation 2 can react with two molecules of carbon dioxide and one molecule of water (Equation 5). The result is azurite, which is a compound that varies in color from shades of blue to shades of purple.
The third reaction that influences the color of patina is when four molecules of copper oxide from Equation 2 reacts with one sulfate molecule and three water molecules (Equation 6). The result is a compound called brochantite, which varies in color from shades of dark green to emerald.
Chemical Reactions Atomically
- Equation 1: 4Cu + O2 → 2Cu2O [red to pink]
- Equation 2: 2Cu2O + O2 → 4CuO [black]
- Equation 3: Cu + S → CuS [black]
- Equation 4: 2CuO + CO2 + H2O → Cu2CO3(OH)2 [“malachite,” dark green to blue]
- Equation 5: 3CuO + 2CO2 + H2O → Cu3(CO3)2(OH)2 [“azurite,” blue to purple]
- Equation 6: 4CuO + SO3 + 3H2O → Cu4SO4(OH)6 [“brochantite,” dark green to emerald]
Reasons Why Patina Can Be Green and Blue
Patina has different colors and patterns that depend on the unique experience that each copper object has had. The next section explains the physics and chemistry of why the colors of patina can vary so much from place to place and from time to time. This section describes in broad terms why the presence of certain chemical compounds can create green patina on copper and blue copper patina.

Patina Colors from Equations 4-6
- Malachite: dark green to blue
- Azurite: blue to purple
- Brochantite: dark green to emerald

Depending on what combination of those three is present, the color of the copper patina can vary from shades of green to shades of blue. Darker greens indicate an abundance of malachite and brochantite, while blues suggest that azurite is abundant. Each of these three compounds can form beautiful crystalline structures on their own, like precious rocks. When mixed in small amounts and held on the surface of copper, these three compounds produce the pleasant luster of patina.
It is interesting to note that human blood is red because the molecule that carries oxygen in our blood is iron, which produces a red color. Shellfish, such as crabs, have greenish-blue blood because the metal atom that carries oxygen in their bodies is copper.
Factors That Affect Patina Formation
Why does copper patina over time? As we have established, patina forms on copper because of chemical reactions. The faster a chemical reaction is, the more products will form. Several factors in the copper and the surrounding environment can affect how fast patina forms, and what colors it will have.

Knowing these factors will not only help you appreciate the patina on your copper but will also allow you to influence how the patina forms. This knowledge will surely impress your friends and relatives as they marvel at the elegance of your copper and the sophistication it brings to both your home and your conversations.
Temperature
Chemical reactions happen when molecules move and collide with each other. The molecules are too small to see with the naked eye, but we know that chemical reactions happen thanks to a variety of signs, such as a change in color. Heat causes molecules to move faster, whether those molecules are bouncing around in the air or a liquid. Higher temperatures cause more reactions to happen at the surface of the copper.

The higher the temperature, the faster chemical reactions can occur, like in your copper hood. Faster-moving molecules move with more energy, so their collisions with each other are more likely to break connections within a molecule. More than that, high-energy collisions help to form bonds between two molecules. This is similar to how it is easier to dissolve sugar in hot tea than it is to do so in iced tea. Ever wonder why the sugar always sits at the bottom of your iced tea, even though you have been stirring it for five minutes?
Geographic Location
Different regions have different climates. Inland and desert areas can be very hot, while coastal regions are cold. Regions at high elevations are colder than valleys. Often the location of where copper patina sheets are located within your home determines the temperatures that your copper is exposed to.
Room Ventilation and Nearby Sources of Heat
Rooms without windows and ventilation can become very hot during the summer. Stoves, fireplaces, furnaces, heaters and refrigerators produce heat. This heat can warm up the entire room or nearby copper objects like your copper hood or copper sink.

Humidity & Precipitation
The chemical reactions that form patina on copper require the presence of water. Water exists in the form of moisture in the air, mist, rain, and snow. Some places have high humidity, meaning there is a lot of moisture in the air. Some regions have both high temperatures and high humidity, which promotes the chemical reactions that form the patina. Some regions are very dry, so there is minimal natural water vapor in the air.
Precipitation Rate
Different regions receive different amounts of rain and snow. Copper objects exposed to wet weather are frequently drenched or moist. Some regions have rain for most of the year, while others receive only a few weeks of rain per year.

Nearby Sources of Steam
Household appliances can produce steam, which moistens the air. Kettles, water boilers, and pressure cookers release significant amounts of steam that can affect nearby copper objects or ones in the same room.
Impurities in the Copper
Copper ore taken from the ground has many impurities in it. Through the process of electrolysis, pure copper is obtained. However, copper can contain impurities in the form of other metals, such as iron, nickel, cobalt and molybdenum. These impurities can also react with oxygen and water vapor, just like copper does, to form compounds that have color.
Iron can form iron oxides that have a reddish brown color. Cobalt can form cobalt oxides that have a greenish-brown color. The amount of impurities in the copper can affect the color of your copper and patina sink and other copper products.
Air Pollution
In Equation 6 above, copper oxide reacts with sulfur oxide and water to form brochantite, which gives a greenish color to patina. Sulfur exists in the atmosphere in the form of sulfur oxide. One source of sulfur oxide air pollution is through burning fossil fuels, such as oil and gasoline. In fact, acidic rain that damages buildings and statues gets its acidity in part from the sulfur oxides that mix with water vapor to form sulfuric acid.
Urban areas that have many automobiles will have high amounts of sulfur oxides in the atmosphere. Other sources of sulfur oxide production in the air are industrial regions that release smoke, such as power plants and chemical production plants. All of these things affect the color of copper patina.
Copper Patina Throughout History
Copper history is fascinating and you can see the result of copper in history in buildings that still exist today. Once you’ve learned how to recognize its distinct green appearance, you may be surprised at how common it is.
The Statue of Liberty might be the most famous copper patina in the world. It’s hard to imagine that it was brown when it was first assembled! Here are a few more buildings that feature copper patinas in various shades of green:
Berlin Cathedral

The Berlin Cathedral is one of the most recognizable buildings in Berlin. It was built between 1894 and 1905, and features five copper domes and multiple copper statues. Although it was damaged in World War II, it has since been restored, and retains its iconic green patina.
Nordic Embassies, Berlin

Also located in Berlin, the Nordic Embassies are another unique example of copper in architecture, this time with a distinctly modern appearance. It contains buildings representing each of the Nordic Countries, surrounded by a green copper wall that winds its way around the complex. According to its architect, “the copper strip is a sequence of 3,850 copper lamellas of the same type.” Completed in 1999, the wall provides a cohesive look to the buildings, with the lamellas inclined at different angles to allow for light and air to pass through.
Belvedere Palace, Vienna

The Belvedere Palace in Vienna is an example of copper as used in Baroque architecture. Built in the early 1700s, the copper roof is still intact 300 years later and looks great today. Allegedly, the design was inspired by the shape of Ottoman army tents camped outside the city of Vienna.
Temppeliauko Church, Helsinki

The Temppeliauko Church in Helsinki, Finland, is another stunning piece of modern architecture. Completed in 1969, this church was excavated out of solid granite bedrock, and features a large copper dome surrounded by 180 window panes that act as a skylight.
The church is known for its great acoustics, and has an organ with 3001 pipes! The copper adds to the rugged, cave-like interior, while its patina-covered exterior is visible on the top of the hill.
Jurassic Museum of Asturias, Spain

Located in Northern Spain on the Atlantic coast, the Jurassic Museum of Asturias is designed in the shape of a dinosaur’s footprint. The roof is made of a plywood frame covered with a copper plate to protect it from the marine air. Since the building was only opened in 2004, you can see that the roof is still primarily its original brownish copper color, and is slowly developing a green patina.
Minneapolis City Hall

The Minneapolis City Hall was built between 1888 and 1909, and originally had a terra cotta roof. But when it started leaking, it was replaced with a copper roof instead. At the time it was installed, it was the largest and heaviest roof in the U.S., weighing over 180,000 pounds!
Fairmont Le Chateau Frontenac, Montreal

The Chateau Frontenac is one of the most iconic buildings in Quebec, Canada. Because part of the roof was replaced in 2011-2012, it’s easy to see the difference between the original roof, which still has its green patina, and the renovated roof, which now has a shiny brown color.
Peckham Library, London

Finally, let’s take a look at the Peckham Library, a prize-winning building that opened in London in 2000. The exterior of the building is constructed with pre-patinated copper, and has a distinct bright green appearance, complemented with steel mesh and colored glass windows. Part of the building is elevated above the street to create a pedestrian area and reduce noise from street-level traffic.
How Patina Develops
When designing with copper, it’s important to have a vision not just of how it will look today, but what it will look like in the future. Not all patina develops at the same rate, and the ultimate appearance of the patina depends on the environment.
Buildings located in industrial environments will develop a copper sulfide patina due to acid rain, while buildings in rural areas will develop a basic copper carbonate patina. Copper in wet marine environments, such as the Jurassic Museum, can expect to develop a patina more quickly. Some exteriors are clad with copper alloys, rather than pure copper, which weather at a different rate.
You Can Create Patina
If you don’t want to wait for an aquired patina to develop, you can create an applied patina using chemical processes. A simple vinegar and salt mixture can create an applied patina on a piece of copper jewelry or a decorative item. An instant patina isn’t suitable for use in architecture, but it might be long-lasting enough for an art project or an interior design feature.
Copper and patina may seem to go hand in hand,but copper isn’t the only material to aquire a patina. Carbon steel woks, terra cotta pots, and wooden furniture items can all develop patinas, although they each have their own appearance. For example, Japanese knives are traditionally made using a process called nekonoshoben, which uses an acidic solution to produce a protective blueish patina on the carbon steel blade.
A metalworker can help you determine the best way to care for your copper patina to provide a consistent look or to help it age gracefully over time.
Types of Patina Available at CopperSmith
At CopperSmith, we do not boast about our excellent selection of beautiful patinas without good reason. We have a range of patina colors to choose from, so you should have no problem finding the right shade that matches your home décor. You do not have to wait years or decades to have the right shade of patina. We provide matured patinas that you can enjoy immediately.
Shades of patina available at CopperSmith include:
- Natural Raw Copper [reddish pink]
- Old Coin [reddish brown]
- Antique [dull brown]
- Dark Antique [dark brown]
- Bronze [dark blue]

Not only do we understand the nuances of different shades of copper patina, but we also add an extra layer of beauty to copper furnishes that the patina itself cannot achieve: texture. The physical texture of the copper gives depth to, and brings the character out of, the patina.
Textures of copper patina available at CopperSmith include:
- Smooth
- Hood hammered
- Beehive hammered
- Light hammered
- Hand hammered

The beauty of copper patina has yet another dimension of depth and elegance that is unique. Just as the hammered texture of copper changes how light reflects off its surface, three-dimensional carvings captivate the imagination by suggesting hidden stories behind each curve. Ornate carvings can imbue a copper item with a charming nature. Alternatively, they can add a dash of regal pride to an otherwise mundane household fixture.
Custom-designed carvings in CopperSmith products include:
- Landscapes
- Gardens
- Royal emblems
- Geometrical patterns
- Birds
- Fish
- Bricks
Custom Copper Fixtures
CopperSmith has copper furnishings for every room in the house: kitchens, bathrooms, dining rooms, bedrooms and even the garage. Browse our website to see all the different copper products that we offer.
Range Hoods
Copper range hoods above the stove can easily become the centerpiece of the kitchen. Copper patina has a presence that will inevitably draw attention from all corners of the room. Decorate your range hood with straps and rivets that themselves can have textured patterns. Not only do range hoods serve the important safety function of removing fumes and particulate matter from the air, but they can also be a pleasure to the eyes.
Sinks
Aside from the stove, the kitchen sink is a quintessential piece of furnishing in the kitchen. CopperSmith has apron sinks and barn sinks that display a face towards the kitchen that draws attention from across the room. Sinks are not just found in the kitchen but are also in the bathroom. Garages can have sinks next to the washer or for everyday use, so even that room can be enhanced by copper. Some people prefer to have copper sinks next to their outdoor grill or in their patio, making food preparation and clean up easier. Check this out if you're interested in learning about the different styles of kitchen sinks.

Tubs
Copper bathtubs have a unique feel of luxury and character. Not only do they have elegant designs, but they are easy to clean, as well.

Tabletops
Copper tabletops are versatile pieces of furniture that can go into any room in the house, whether as a dining table, coffee table or nightstand.

Examples of other copper items at CopperSmith include:
- Light Fixtures
- Mirrors
- Knobs & Pulls
- Towel Bars & Rings
- Backsplash Murals
- Tapestry Hangers
- And more!
How to Create Your Desired Patina
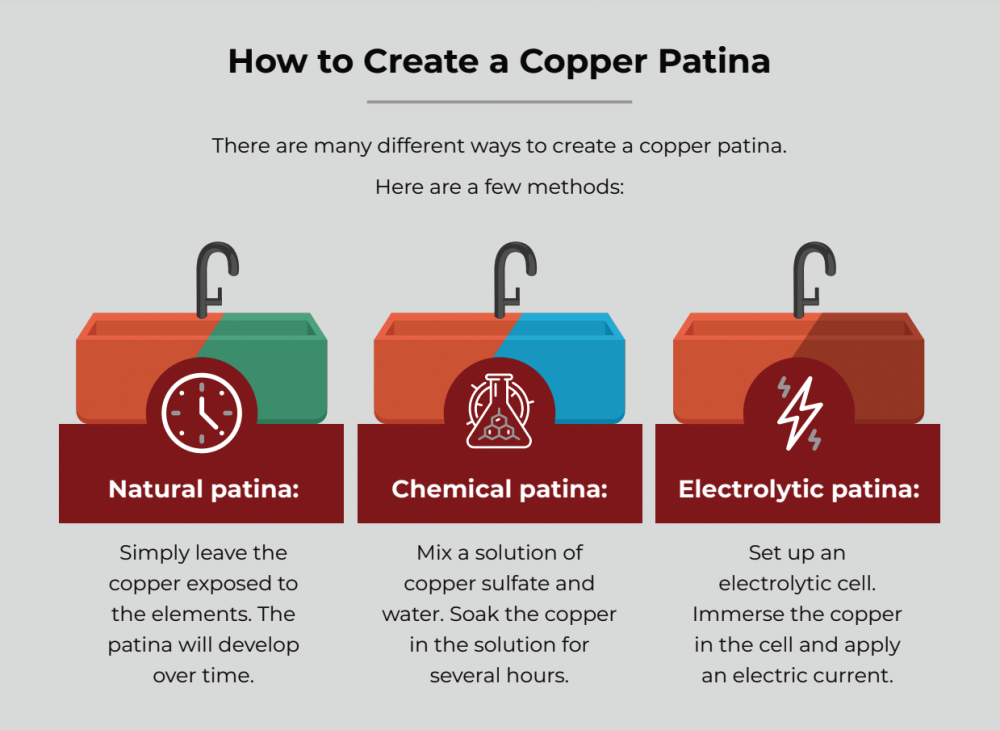

Since we know the chemical reactions that cause the different shades of color in copper patina, there are ways that we can affect what color patina becomes and how fast patina forms. Creating artificial patina is a common activity among people who make copper jewelry and kitchenware. The following tips will teach you how to patina copper.
Making a consistent pattern of artificial patina on large furnishings such as bathtubs, tables and range hoods is more challenging than doing it on jewelry. For large objects, it is best to pick a piece of copper whose patina was industrially created by professionals.
For each of the following methods, the longer the copper is exposed to the homemade concoction of chemicals, the more intense the patina will become. You should be sure to test out the strength of your treatment on small objects such as coins and pennies before doing it on your prized copper possessions.
Keep notes about how much of each chemical was mixed, which will allow you to adjust the strength or effect of your home-made patination mixture for your desired effect. Test the method to see how fast the patina forms, so you know when to stop the incubation time.
Make sure you pay attention to safety procedures about handling any home cleaning agent. An adult should supervise children under 18. Household cleaning agents can cause irritation in the eyes, skin, nasal passage and lungs. They can also be dangerous if ingested.
Incubate with Hot Crushed Boiled Eggs
The hot boiled egg method is perfect for creating a brown patina on copper. Eggs are hard boiled and then crushed to pieces in a re-sealable plastic bag or container while they are still hot. The copper is then placed into the bag and sealed for minutes to hours, depending on the desired shade of brown patina. The longer the incubation time, the darker the brown color. This method is ideal for small copper items, but large ones are difficult to manage through this technique.
Use Miracle-Gro
Miracle-Gro plant fertilizer is an excellent oxidizer of copper. For a blue patina, mix one part Miracle-Gro with three parts water for a solution that you can spray or wipe onto the copper. For a green patina, mix one part Miracle-Gro with three parts red wine vinegar. A patina will form within 30 minutes and become permanent within 24 hours.
Soaking in White Vinegar and Salt, Sawdust or Potato Chips
Soaking copper in white vinegar and salt will create a blue or green patina. Other ways of doing this are to bury the copper in sawdust or crushed potato chips soaked in white vinegar. The longer the copper is buried, the darker the patina becomes.

Expose Copper to Saltwater and Ammonia Vapors
Spray the copper with a solution of salt water and then place the copper in a container that contains non-detergent ammonia. The copper should not be touching the ammonia liquid, but should be raised above it.
The sealed container will trap ammonia vapors that will then make contact with the copper. Be careful when handling ammonia: wear protective gloves, wear safety goggles and be near water in case you need to rinse ammonia off of yourself. Also, do not sniff ammonia and use ammonia outdoors or make sure the room is well-ventilated.
How to Naturally Influence the Color of Patina
There are ways to allow the patina to form naturally without applying chemicals to it. Exposing the copper to outdoor conditions will help patina to form. Outdoor conditions include fog, rain, snow, and heat. Leaving the windows open will change the temperature and moisture content of the room, so any copper objects in that room will also experience the changes.
You can purposely leave copper furnishings next to sources of heat and moisture, such as in the kitchen next to the stove or the back of the refrigerator. Leaving copper next to a window so that it receives lots of sunlight will expose the copper to natural heat.
Sealing the Patina to Keep Your Desired Color
The patina that forms from artificial methods can be easily brushed off, so it must be sealed to be kept permanent. Sealants can be wax, lacquer or a proprietary mixture of chemicals. Jewelry-making and craft stores carry multiple brands of sealants for protecting patina.
Since patina forms through chemical reactions between copper and molecules in the air, preventing the air from reaching the copper will stop any more patina from forming. Applying a sealant can also be a way to keep the patina that you like, preventing it from changing further.
How to Look After Copper Patina

Before you start cleaning your copper, you need to check to see if there is a lacquer finish, as certain cleaning methods will depend on the presence of lacquer. You can clean lacquered copper with water and mild detergent and wipe the residue with a dry cloth.
If you need to find out if your copper is lacquered, you can rub the surface of the copper with a microfiber cloth that has been dipped in baking soda and white vinegar. If the tarnish disappears, your copper is not lacquered. If the item is lacquered, you need to wash it with water and mild dish soap.
How Can You Clean Copper Naturally?
Lots of people worry about using harsh chemicals on their copper. Luckily, there are a number of household ingredients you can use to clean copper naturally:
White Vinegar and Table Salt
You can create a paste with salt and white vinegar, apply it to the surface, use a soft cloth to buff it then rinse and dry. You can also use a toothbrush to scrub any dirt or grime that has collected.
Ketchup
You can apply a layer of ketchup to a copper item and rub the condiment over the surface. The tomato's acidity can help to remove any tarnish. Make sure you rinse and dry the copper afterward.
Lemon Juice
You can use lemon juice in one of two ways:
- Use it with baking soda and turn it into a mixture that you can apply to the copper surface and buff it with a soft and clean cloth.
- You can cut a lemon in half and put table salt into the pulp, which creates a natural sponge. Then, rub the lemon on the patina's surface and use any extra salt necessary to remove any tarnish before rinsing and drying.
There are a number of ways to clean and preserve copper both, physically and chemically, such as water blast cleaning and mechanical abrasive cleaning to remove the corrosion while preserving the patina.
Get Advice on Including Copper Furnishings to Your Home Décor
Your Home Décor
We hope you have found this copper patina guide useful because copper is an amazing material that has been present throughout history. If you are looking for ways to create a striking and stylish home environment, copper and patina should undoubtedly be a big part of it. That's due to its diverse uses in planters, fireplaces, bathtubs, and so much more.
The experts at CopperSmith are waiting to provide expert advice on how to design the copper hoods and copper farmhouse sink furnishings that are perfectly unique to your home and needs. We stand behind our money-back guarantee because we believe in delivering only the best service and copper fire pit products. We can send you samples of our copper patinas so that you can know the exact look and feel of our copper for yourself.
Our expert designers will walk you through the process and options, taking the guesswork out of the shopping and planning process. Our knowledge and experience make it easy for you to get the right furnishing that will have the right fit for every location and purpose.

FAQs
What is copper patina?
Copper patina is a natural film that forms on copper over time because of oxidation, and it can be green, blue, or brown. This film protects your copper product compared to something like rust that forms on iron and steel and weakens it.
Can patina be stopped?
Yes, patina can be stopped if there is a protective sealant applied to the copper sheets used to create your product.
Does copper always turn green?
No, copper does not always turn green, but if it is untreated and exposed to moisture and oxidation, it can form a green patina.
Can I choose my patina colors?
Typically, patina colors are created on patina sheets before creating the final product. But if you would like a customized copper hood, sink, or other product, you can choose the color of the patina.








 By submitting, you agree to our Privacy Policy & Terms
By submitting, you agree to our Privacy Policy & Terms